
FDAQRC is proud to be exhibiting at
the RQA 2023 conference.
FDA Quality and Regulatory Consultants is a leading provider of innovative regulatory compliance solutions for the biotech and pharmaceutical industry. Our internal staff and global network of consultants are experienced professionals with expertise across all GxP areas. FDAQRC is equipped to assist with inspection readiness, GAP analysis, QMS development and more!
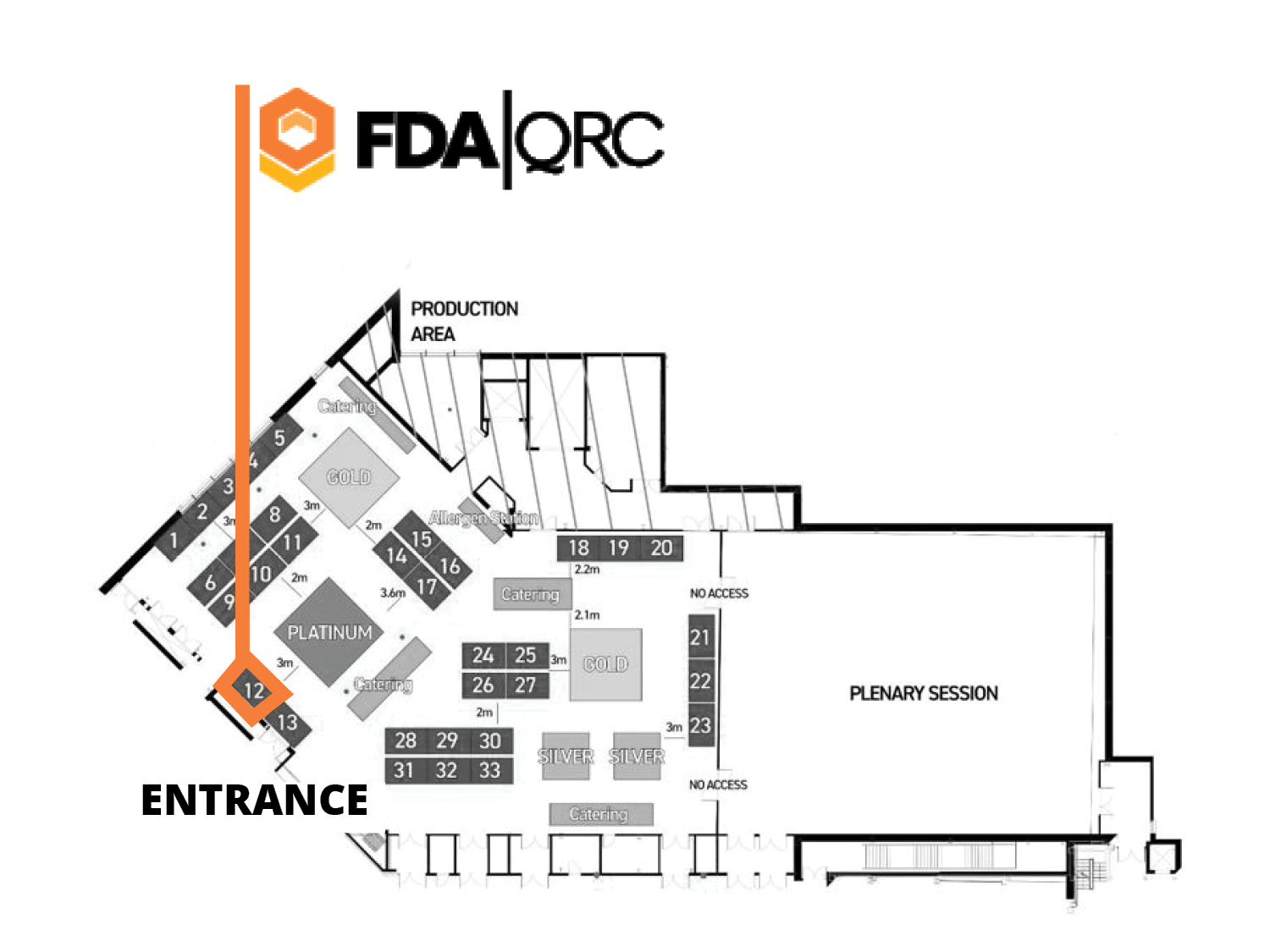
The FDAQRC staff attending RQA 2023 are eager for the opportunity to make new connections with like-minded professionals in our industry. If you would like to discuss how our expertise across GxPs can improve your regulatory compliance and quality assurance processes, come see us at Booth 12 during the conference.
Meet With Us
If you can meet with us at RQA, stop by booth 12 (near the entrance/exit) at any time during the Exhibit Hall hours and ask for Steve Bliss or Chris Rush!
Need to chat at a later time? No worries, fill out our form and a team member will reach out to you to schedule a meeting.
